 
Moreover, the matrix is always positive definite that is to say, the eigenvalues are all strictly positive. In particular, if the set is normalized (though not necessarily orthogonal) then the diagonal elements will be identically 1 and the magnitude of the off-diagonal elements less than or equal to one with equality if and only if there is linear dependence in the basis set as per the Cauchy–Schwarz inequality. Ī quantitative measure of the overlap of two atomic orbitals Ψ A and Ψ B on atoms A and B is their overlap integral, defined as The carbon hybrid orbitals have greater overlap with the hydrogen orbitals, and can therefore form stronger C–H bonds. The dashed lines, each connecting two lobes, indicate the side-by-side. 4: (a) In the acetylene molecule, C2H2, there are two CH bonds and a CC triple bond involving one CC bond and two CC bonds. Two sp 3 carbons can also overlap to form a CC sigma bond where two sp 3 orbitals overlap head to head, such as in the formation of the. The two carbon atoms of acetylene are thus bound together by one bond and two bonds, giving a triple bond. Pauling proposed that s and p orbitals on the carbon atom can combine to form hybrids (sp 3 in the case of methane) which are directed toward the hydrogen atoms. The bonds between the sp 3 orbitals of hybridized carbon and the s orbitals of hydrogen in methane are also example of sigma bonds. As s orbitals are spherical (and have no directionality) and p orbitals are oriented 90° to each other, a theory was needed to explain why molecules such as methane (CH 4) had observed bond angles of 109.5°. Linus Pauling explained the importance of orbital overlap in the molecular bond angles observed through experimentation it is the basis for orbital hybridization. Orbital overlap can lead to bond formation. In chemical bonds, an orbital overlap is the concentration of orbitals on adjacent atoms in the same regions of space. Nevertheless, it is very easy to determine the state of hybridization and geometry if we know the number of sigma bonds and lone pairs on the given atom. They are used to make one sigma bond with the carbon and contain three lone pairs.Concentration of chemical orbitals on adjacent atoms * The chlorine atoms are hybridised sp 3: it has four orbitals sp 3. The 2p z orbital make one pi bond with the 2p z from the carbon. The sp 2 orbitals form one sigma bond with the carbon and contain two lone pairs. It has three orbitals sp 2 and one orbital 2p z. * The oxygen atom is also hybridised sp 2. The 2p z orbital make one pi bond with the oxygen atom. 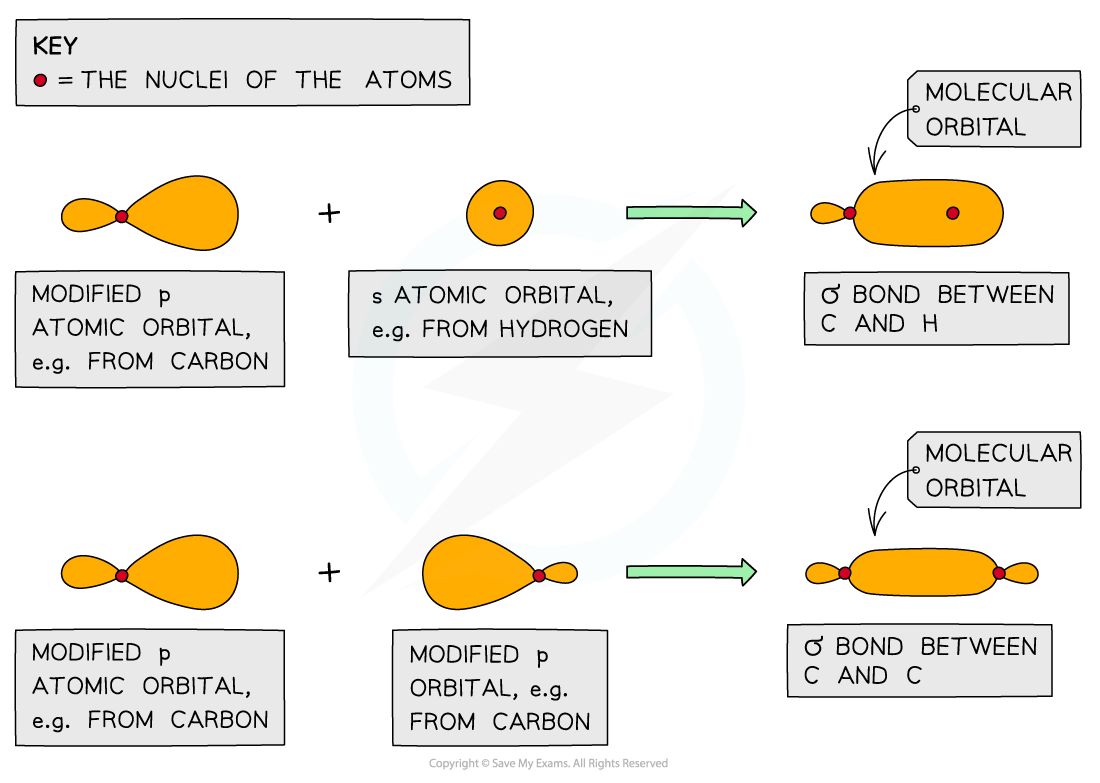
It form three sigma bonds with the two Cl and O atoms, using the sp 2 orbitals. As a consequence, there is one 2p orbital left unhybridised and available to make one pi bond. The name sp 2 means that the one orbital 2s and two orbitals 2p mixed to form three hybridised sp 2 orbitals. As a consequence, two 2p orbitals are left unhybridised and available to make two pi bonds. The name sp means that the orbital 2s and only one orbital 2p mixed to form two hybridised sp orbitals. 1.10, 1.17, 1.18 in text) A sigma bond results from the overlap between an s orbital and any other atomic orbital. Then, the angles expected and observed agree (180º for sp, 120º for sp 2 and 109.5º for sp 3). A sigma bond can be formed in a variety of ways. It consists in mixing 2s and 2p orbitals together at the beginning to form new sp, sp 2 or sp 3 orbitals (depending on the case). Lone pairs occupy the hybridized orbitals. Step 2: Determine the hybridization of any atoms with lone pairs (heteroatoms). Step 1: Add any missing lone pair electrons to the heteroatoms (atoms other than carbon and hydrogen). In order to explain this difference of angles between the expected 90º and the observed angles, the theory of hybridisation was proposed. To identify the orbitals of the lone pair electrons in non-aromatic compounds, we can follow a two-step approach. This means that sp hybridized atoms can form only sigma bonds. Hybridization(72) Molecular Orbital Theory(37) MO Theory: Homonuclear Diatomic. sp-hybridized atoms use all three p orbitals for the hybridization. Lewis Dot Structures: Sigma & Pi Bonds(25) Lewis Dot Structures: Ions(11). 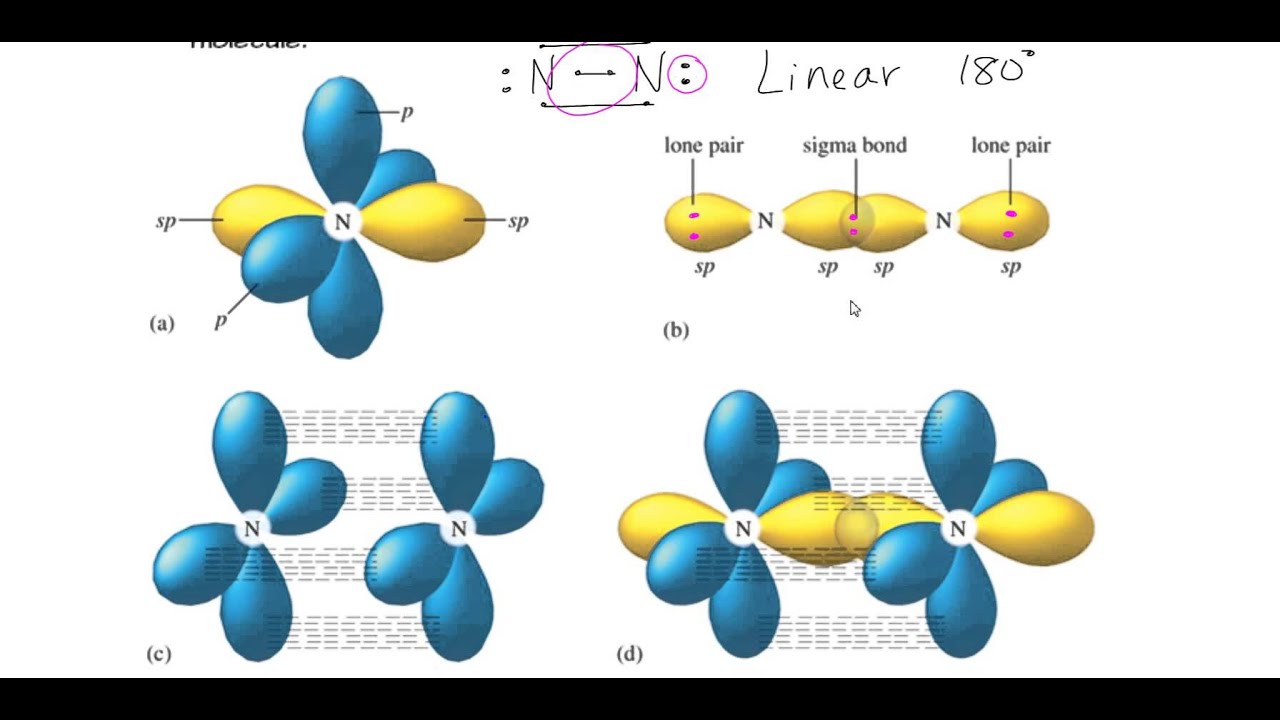
They are closer to: 109.5º, 120º and 180º. When two hybridized orbitals Orbital overlap - Wikipedia, they form a bond. * However, when that carbon atom is place inside a molecule, the angles observed are never 90º. When two p orbitals overlap side-to-side, they form a bond. When two s orbitals overlap, they form a bond. So, the angles between the 2p orbitals are 90º. Explanation: An atoms valence shell has one s orbital and three p orbitals. The 2s is spherical and the 2p have two lobes pointing towards one direction: x, y and z. Q.24 Considering the quadrupolar nature of M-M bond in Re2C182, the M-M bond order. * An isolated carbon atom possesses four atomic orbitals: 2s, 2p x, 2p y and 2p z. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
